Phytochemical Insights into Rhizophora stylosa: pH-Dependent Extraction, Antioxidant Activity, and Enzyme Inhibition Potential
Abstract
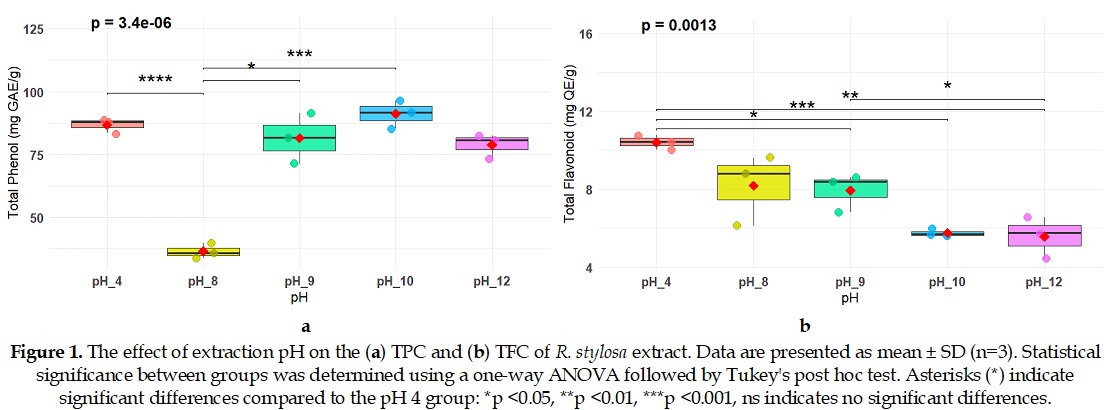
The solvent properties, including pH, highly influence the extraction efficiency of these bioactive compounds. This study investigated the effect of pH modulation in the ethanol solvent on the recovery of antioxidants from Rhizophora stylosa. Ethanolic extraction was performed on R. stylosa samples using solvents adjusted to varying pH levels (4, 8, 9, 10, and 12). The total phenolic content (TPC) and total flavonoid content (TFC) of the extracts were quantified using standard colorimetric assays. The antioxidant activity was evaluated using the DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging assay. The inhibitory activity against carbohydrate-digesting enzymes was assessed through α-amylase and α-glucosidase inhibition assays. Additionally, data mining of established natural product databases (KNApSAcK and IMPPAT) and literature was conducted to identify and catalog the known bioactive constituents of R. stylosa. The results demonstrated a significant correlation between the pH of the extraction solvent and the yields of phenolic compounds, flavonoids, and the resulting antioxidant activity. Extract obtained at pH 10 yielded the highest TPC and exhibited the strongest antioxidant activity, whereas the highest TFC was observed at pH 4. Furthermore, enzymatic inhibition assays revealed that the pH 10 extract displayed moderate α-amylase inhibitory activity (IC₅₀ = 76.7 µg/mL) but substantially weaker α-glucosidase inhibition (IC₅₀ = 190 µg/mL), demonstrating a selective inhibition profile though less potent than that of acarbose. These findings establish pH-modulated extraction as a targeted strategy to produce potent, phenolic-rich antioxidant extracts from R. stylosa, highlighting its potential for nutraceutical and pharmaceutical applications.
Full text article
References
2. Wahyuni S, Mahmuda INN, Maryati, Perdana AP, Maharotullaili NA. Studi Komparatif Invitro Aktivitas Antioksidan Ekstrak Etanol, Fraksi Etil Asetat Dan Isolat Aktif Kulit Delima (Punica Granatum L.). Biomedika. 2023;15(1):67-75. DOI: 10.23917/biomedika.v15i1.1751.
3. Lin W, Li G, Xu J. Bio-Active Products from Mangrove Ecosystems. Mar Drugs. 2023;21(4):239. DOI: 10.3390/md21040239. PMID: 37103378; PMCID: PMC10145032.
4. Muhtadi M, Pambudi DB, Maryati M. Mechanistic Insight into Medicinal Properties of Indonesian Diverse Mangrove Species: A Review. Int J Appl Pharm. 2024;16(5):1-8. DOI: 10.22159/ijap.2024v16s5.52488.
5. Miranti DI, Ichiura H, Ohtani Y. The Bioactive Compounds and Antioxidant Activity of Food Products of Rhizophora stylosa Fruit (Coffee and Tea Mangrove). Int J For Res. 2018;2018(1):2315329. DOI: 10.1155/2018/2315329.
6. Wang Y, Zhu H, Tam NFY. Polyphenols, tannins and antioxidant activities of eight true mangrove plant species in South China. Plant Soil. 2014;374(1–2):549-63. DOI: 10.1007/s11104-013-1912-9.
7. Ahmed S, Alsharif KF, Aschner M, Alzahrani KJ, Akkol EK, Türkcanoğlu EG, et al. A deep dive into herbal extraction: Techniques, trends, and technological advancements. S Afr J Bot. 2026;188:9-37. DOI: 10.1016/j.sajb.2025.11.005. PMID: 41716686; PMCID: PMC12916026.
8. Heck N, Goldberg L, Andradi-Brown DA, Campbell A, Narayan S, Ahmadia GN, et al. Global drivers of mangrove loss in protected areas. Conserv Biol. 2024;38(6):e14293. DOI: 10.1111/cobi.14293. PMID: 38766900; PMCID: PMC11589005.
9. Rajković KM, Vasić M, Drobac M, Mutić J, Jeremić S, Simić V, et al. Optimization of extraction yield and chemical characterization of optimal extract from Juglans nigra L. leaves. Chem Eng Res Des 2020;157:25–33. DOI: 10.1016/j.cherd.2020.03.002.
10. Cabrera MN, Rossi A, Guarino JI, Felissia FE, Area MC. Alkaline Extraction and Ethanol Precipitation of High-Molecular-Weight Xylan Compounds from Eucalyptus Residues. Polymers. 2025;17(12):1589. DOI: 10.3390/polym17121589. PMID: 40574117; PMCID: PMC12196941.
11. Csuti A, Zheng B, Zhou H. Post pH-driven encapsulation of polyphenols in next-generation foods: principles, formation and applications. Crit Rev Food Sci Nutr. 2024;64(33):12892-906. DOI: 10.1080/10408398.2023.2258214. PMID: 37722872.
12. Yulyana A, Chaidir C, Simanjuntak P, Sulastri L, Abdillah S. The water fraction of Cantigi (Vaccinium varingiaefolium Bl. Miq.) fruits demonstrate the highest antimetabolic syndrome properties on enzyme assay. Pharmacia. 2023;70(3):587–94. DOI: 10.3897/pharmacia.70.e109333.
13. Fajriyah NN, Muhtadi M, Rosyidand FN, Mugiyanto E. Characterization of nano-hydrogel chitosan-annona muricata extract by using ionic gelation. AIP Conf Proc. 2024;3070:020008. DOI: 10.1063/5.0198874.
14. Pérez M, Dominguez-López I, Lamuela-Raventós RM. The Chemistry Behind the Folin-Ciocalteu Method for the Estimation of (Poly)phenol Content in Food: Total Phenolic Intake in a Mediterranean Dietary Pattern. J Agric Food Chem. 2023;71(46):17543-53. DOI: 10.1021/acs.jafc.3c04022. PMID: 37948650; PMCID: PMC10682990.
15. Muhtadi M, Haryoto H, Sujono TA, Suhendi A. Antidiabetic and Antihypercholesterolemia Activities of Rambutan (Nephelium lappaceum L.) and Durian (Durio zibethinus Murr.) Fruit Peel Extracts. J Appl Pharm Sci. 2016;6(4):190–4. DOI: 10.7324/JAPS.2016.60427.
16. Shraim AM, Ahmed TA, Rahman MM, Hijji YM. Determination of total flavonoid content by aluminum chloride assay: A critical evaluation. LWT. 2021;150:111932. DOI: 10.1016/j.lwt.2021.111932.
17. Baliyan S, Mukherjee R, Priyadarshini A, Vibhuti A, Gupta A, Pandey RP, et al. Determination of Antioxidants by DPPH Radical Scavenging Activity and Quantitative Phytochemical Analysis of Ficus religiosa. Molecules. 2022;27(4):1326. DOI: 10.3390/molecules27041326. PMID: 35209118; PMCID: PMC8878429.
18. Mugiyanto E, Cahyanta AN, Putra I, Setyahadi S, Simanjuntak P. Identifying active compounds of soursop ethanolic fraction as α-glucosidase inhibitor. Pharmaciana. 2019;9(2):191–200. DOI: 10.12928/pharmaciana.v9i2.10105.
19. Zapata F, López-Fernández A, Ortega-Ojeda F, Quintanilla G, García-Ruiz C, Montalvo G. Introducing ATR-FTIR Spectroscopy through Analysis of Acetaminophen Drugs: Practical Lessons for Interdisciplinary and Progressive Learning for Undergraduate Students. J Chem Educ. 2021;98(8):2675-86. DOI: 10.1021/acs.jchemed.0c01231. PMID: 35281766; PMCID: PMC8908246.
20. Zhu J, Chen C, Zhang B, Huang Q. The inhibitory effects of flavonoids on α-amylase and α-glucosidase. Crit Rev Food Sci Nutr. 2020;60(4):695-708. DOI: 10.1080/10408398.2018.1548428. PMID: 30638035.
21. Rauf A, Khan IA, Muhammad N, Al-Awthan YS, Bahattab O, Israr M, et al. Phytochemical composition, in vitro urease, α-glucosidase and phosphodiesterase inhibatroy potency of Syzygium cumini (Jamun) fruits. S Afr J Bot. 2021;143:418–21. DOI: 10.1016/j.sajb.2021.04.006.
22. Afendi FM, Okada T, Yamazaki M, Hirai-Morita A, Nakamura Y, Nakamura K, et al. KNApSAcK family databases: integrated metabolite-plant species databases for multifaceted plant research. Plant Cell Physiol. 2012;53(2):e1. DOI: 10.1093/pcp/pcr165. PMID: 22123792.
23. Mohanraj K, Karthikeyan BS, Vivek-Ananth RP, Chand RPB, Aparna SR, Mangalapandi P, et al. IMPPAT: A curated database of Indian Medicinal Plants, Phytochemistry And Therapeutics. Sci Rep. 2018;8(1):4329. DOI: 10.1038/s41598-018-22631-z. PMID: 29531263; PMCID: PMC5847565.
24. Chen X, Zhai R, Li Y, Yuan X, Liu ZH, Jin M. Understanding the structural characteristics of water-soluble phenolic compounds from four pretreatments of corn stover and their inhibitory effects on enzymatic hydrolysis and fermentation. Biotechnol Biofuels. 2020;13:44. DOI: 10.1186/s13068-020-01686-z. PMID: 32175010; PMCID: PMC7065323.
25. Zhong X, Zhang S, Wang H, Yang J, Li L, Zhu J, et al. Ultrasound-alkaline combined extraction improves the release of bound polyphenols from pitahaya (Hylocereus undatus 'Foo-Lon') peel: Composition, antioxidant activities and enzyme inhibitory activity. Ultrason Sonochem. 2022;90:106213. DOI: 10.1016/j.ultsonch.2022.106213. PMID: 36327918; PMCID: PMC9636185.
26. Zhang L, Wang Y, Cao Y, Wang F, Li F. Review: Enhancing the Bioavailability and Stability of Anthocyanins for the Prevention and Treatment of Central Nervous System-Related Diseases. Foods. 2025;14(14):2420. DOI: 10.3390/foods14142420. PMID: 40724240; PMCID: PMC12294332.
27. Ilie EI, Popescu L, Luță EA, Biță A, Corbu AR, Mihai DP, et al. Phytochemical Characterization and Antioxidant Activity Evaluation for Some Plant Extracts in Conjunction with Pharmacological Mechanism Prediction: Insights into Potential Therapeutic Applications in Dyslipidemia and Obesity. Biomedicines. 2024;12(7):1431. DOI: 10.3390/biomedicines12071431. PMID: 39062004; PMCID: PMC11274650.
28. Shi L, Zhao W, Yang Z, Subbiah V, Suleria HAR. Extraction and characterization of phenolic compounds and their potential antioxidant activities. Environ Sci Pollut Res Int. 2022;29(54):81112-29. DOI: 10.1007/s11356-022-23337-6. PMID: 36201076; PMCID: PMC9606084.
29. Shahidi F, Samarasinghe A. How to assess antioxidant activity? Advances, limitations, and applications of in vitro, in vivo, and ex vivo approaches. Food Prod Process Nutr. 2025;7(1):50. DOI: 10.1186/s43014-025-00326-z. PMID: 41178997; PMCID: PMC12572074.
30. Esmaeili AK, Taha RM, Mohajer S, Banisalam B. Antioxidant Activity and Total Phenolic and Flavonoid Content of Various Solvent Extracts from In Vivo and In Vitro Grown Trifolium pratense L. (Red Clover). Biomed Res Int. 2015;2015:643285. DOI: 10.1155/2015/643285. PMID: 26064936; PMCID: PMC4438149.
31. Li J, Chen L, Chu X, Ba K, Xie T, Yan W, et al. Synergistic Modulation of π-π* and n-π* Transitions by In Situ Phenol-Like Structure Integration for Efficiently Wide-Spectrum Hydrogen Production of Ultrathin Carbon Nitride. Small. 2024;20(46):e2405013. DOI: 10.1002/smll.202405013. PMID: 39109579.
32. Wagner-Wysiecka E, Łukasik N, Biernat JF, Luboch E. Azo group(s) in selected macrocyclic compounds. J Incl Phenom Macrocycl Chem. 2018;90(3):189-257. DOI: 10.1007/s10847-017-0779-4. Erratum in: J Incl Phenom Macrocycl Chem. 2018;90(3):259. DOI: 10.1007/s10847-018-0788-y. PMID: 29568230; PMCID: PMC5845695.
33. Hawryl A, Hawryl M, Chernetskyy M, Winiarski WW, Oniszczuk A. Integrated Chemometric Assessment, Antioxidant Potential, and Phytochemical Fingerprinting of Selected Stachys and Betonica Plants. Compounds. 2026;6(1):14. DOI: 10.3390/compounds6010014.
34. Chukwuejim S, Kadam D, Aluko RE. Structural, physicochemical, and functional properties of white and blue lupin vicilin and legumin fractions. Food Chem X. 2025;31:103078. DOI: 10.1016/j.fochx.2025.103078. PMID: 41080120; PMCID: PMC12514543.
35. Purwanti N, Roni RAZ, Hakeki AZ, Setiarto RHB. Effects of extraction solvent and isoelectric point on the quality of jack bean (Canavalia ensiformis) protein. Food Sci Biotechnol. 2024;34(6):1317-25. DOI: 10.1007/s10068-024-01795-7. PMID: 40110416; PMCID: PMC11914563.
36. Han X, Wang P, Zhang J, Lv Y, Zhao Z, Zhang F, et al. α-Glucosidase Inhibition Mechanism and Anti-Hyperglycemic Effects of Flavonoids from Astragali Radix and Their Mixture Effects. Pharmaceuticals. 2025;18(5):744. DOI: 10.3390/ph18050744. PMID: 40430562; PMCID: PMC12114633.
37. Assefa ST, Yang EY, Chae SY, Song M, Lee J, Cho MC, et al. Alpha Glucosidase Inhibitory Activities of Plants with Focus on Common Vegetables. Plants. 2019;9(1):2. DOI: 10.3390/plants9010002. PMID: 31861279; PMCID: PMC7020213.
38. Kashtoh H, Baek KH. Recent Updates on Phytoconstituent Alpha-Glucosidase Inhibitors: An Approach towards the Treatment of Type Two Diabetes. Plants. 2022;11(20):2722. DOI: 10.3390/plants11202722. PMID: 36297746; PMCID: PMC9612090.
39. Huong PT, Diep CN, Thanh NV, Tu VA, Hanh TH, Cuong NT, et al. A new cycloartane glucoside from Rhizophora stylosa. Nat Prod Commun. 2014;9(9):1255-7. DOI: 10.1177/1934578X1400900909. PMID: 25918786.
40. Yang XH, Li HB, Chen H, Li P, Ye BP. [Chemical constituents in the leave of Rhizophora stylosa L and their biological activities]. Yao Xue Xue Bao. 2008;43(9):974-8. Chinese. PMID: 19048793.
41. Takara K, Kuniyoshi A, Wada K, Kinjyo K, Iwasaki H. Antioxidative flavan-3-ol glycosides from stems of Rhizophora stylosa. Biosci Biotechnol Biochem. 2008;72(8):2191-4. DOI: 10.1271/bbb.80065. PMID: 18685199.
42. Li DL, Li XM, Peng ZY, Wang BG. Flavanol derivatives from Rhizophora stylosa and their DPPH radical scavenging activity. Molecules. 2007;12(5):1163-9. DOI: 10.3390/12051163. PMID: 17873850; PMCID: PMC6149341.
43. Fahsi N, Mahdi I, Annaz H, Bitchagno GTM, Mahmoud MF, Sobeh M. Unlocking the therapeutic potential of cinchonains: a comprehensive review. Phytochem Rev. 2025;24(1):197–233. DOI: 10.1007/s11101-024-09949-5.
Authors
Copyright (c) 2026 Dwi Bagus Pambudi, Muhtadi Muhtadi, Maryati Maryati

This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
Authors continue to retain the copyright to the article if the article is published in the Borneo Journal of Pharmacy. They will also retain the publishing rights to the article without any restrictions.
Authors who publish in this journal agree to the following terms:
- Any article on the copyright is retained by the author(s).
- The author grants the journal the right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share work with an acknowledgment of the work authors and initial publications in this journal.
- Authors can enter into separate, additional contractual arrangements for the non-exclusive distribution of published articles (e.g., post-institutional repository) or publish them in a book, with acknowledgment of their initial publication in this journal.
- Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their websites) prior to and during the submission process. This can lead to productive exchanges and earlier and greater citations of published work.
- The article and any associated published material are distributed under the Creative Commons Attribution-ShareAlike 4.0 International License.